 If the temperature were to rise to 60☏, a properly functioning float gauge would rise to 40%, assuming the tank was empty at the time of delivery. Using the information from above, a 100 gallon delivery on a cold day (far below 60☏) may indicate less than 100 gallons delivered just by looking at the gauge. Propane users can become quite confused during periods of cold weather following a gas delivery because their propane tank gauge may read less than what they expect it to read. Propane is a liquid and is subject to the same rules of mother nature. As temperatures rise, liquids become less dense and expand. As temperatures fall, liquids become more dense and compact. If the temperature were to rise by the same respective amount, the gauge would indicate a higher volume of propane but there would still be 424 pounds of propane in the tank. The amount of usable energy has not decreased. Although the propane volume has decreased, the amount of propane has not decreased, it has simply become more compact (dense). Assuming the gauge dial sits between 35% and 40% following the temperature drop, there are still 424 pounds of propane in the tank. If there is a significant temperature drop (over 20☏) the gauge will indicate that there is less propane in the tank. 100 gallons of propane weighs 424 pounds (4.24 lbs.A properly functioning float gauge will read 40%.The industry standard 60☏ is universally recognized as the base reference point for liquid propane volume correction. 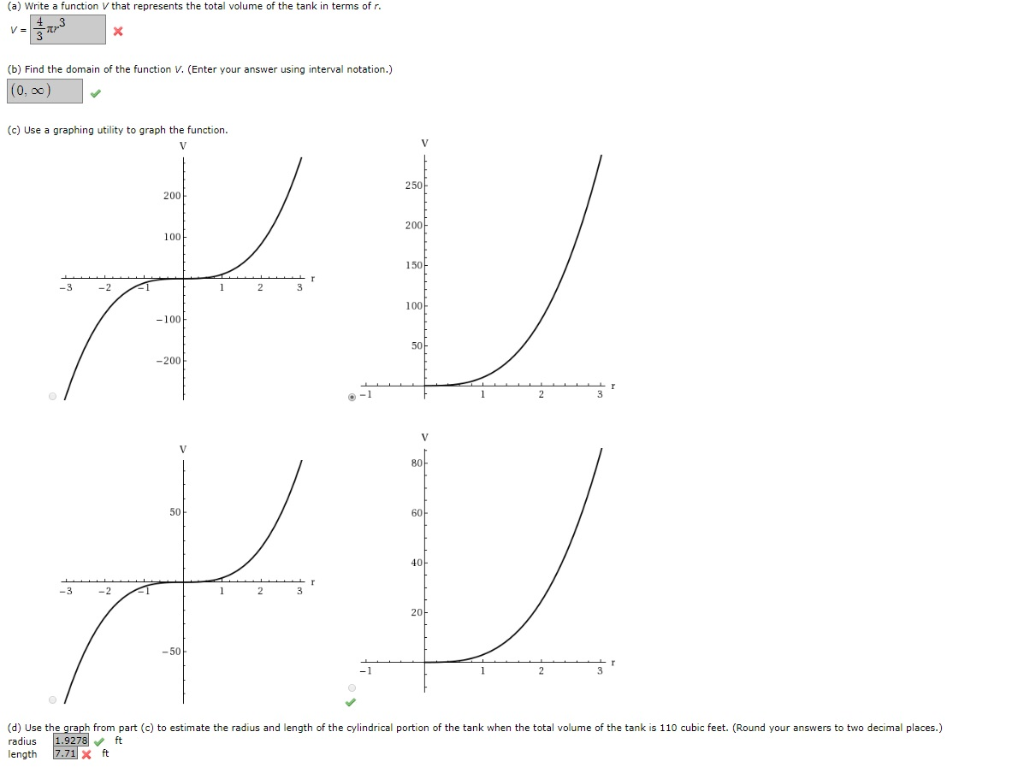
The following example assumes a 250 gallon propane tank has 100 gallons of propane at 60☏. To understand what's going on with regard to differing tank gauge readings in extreme temperatures (hot or cold), we need to first explain basic principles that affect the liquid propane volume. The volume of any liquid will rise and fall dependent upon temperature. In comparison to other liquids regarding temperature and volume, it is no different. This is extremely important to remember when understanding propane temperature and volume and the relationship between the two. Propane is stored and delivered as a liquid. Being temperature dependent, propane becomes more dense as temperature falls and expands as temperature increases. Propane volume rises as temperature rises and falls as temperature falls. In fact, the volume of any liquid in any container is directly related to its temperature. The liquid propane volume in any LP Gas container is directly related to its temperature. Obviously, if your ending temperature equals your starting temperature, the equation simplifies even further.Propane Volume and Temperature Correction This equation can be solved for $m_2$, which is the total mass to add to the tank (which includes the original 1 kg of gas). \fracĪssuming that your tank is "rigid" (not expandable) such that its volume doesn't change, this equation reduces to In addition, if you divide the equation for the initial condition by the equation for the final condition, everything that remains constant drops out of the equation. This equation applies for both the initial condition, and the final condition. Since $$ is the mass of the gas in the tank, substitute "m". 
Thus, the ideal gas law must be modified to account for mass, such that moles are multiplied by molar mass (or molecular weight - MW). Note that the ideal gas law works with moles, and you specified a mass. Obviously, you don't know the volume of the tank, but there is a way to eliminate this variable from the problem. This same equation can be used for the second case, where you pump more gas into the tank in order to achieve a final pressure that contains the desired mass of gas. This equation can be used for the initial case, where you pump 1 kg of gas into the tank and measure the pressure and temperature. Thanks!Įdit2: I could also get a density reading from the mass flow meter, if that would help.įrom the ideal gas law, with the compressibility factor correction for non-ideality, you have the equation $PV=znRT$. 
If someone could find the time to help me out. I converted the pressure to Pa and ran some calculations but didnt get anywhere. $P_1$ - Pressure after $m_1$ was added - 48,87 bar 
Is there a way to accurately calculate the mass needed to fill the tank?Įdit: I forgot to mention, that the tank may not be empty at the beginning. The gas composition is known and i also know the pressure and temperature. I am also able to calculate the compressibility factor z. Then a second point is recorded.įrom there i would need to calculate how much more gas I need to fill, to reach the target pressure. Then the tank is filled with 1 kg of gas (by mass flow meter). I just need to make sure that the tank reaches the right pressure and thus the right density.Ī hose is connected to a tank. I need to calculate the exact amount a tank can take in gas. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
